FAQs
Frequently Asked Questions—
Stem Cell Therapy For Autism
*These statements have not been evaluated by the FDA. Innate Healthcare Institute is a private clinic and any treatments utilizing any form of regenerative medicine is considered experimental.
What does the stem cell collection process look like and what are stem cells doing?
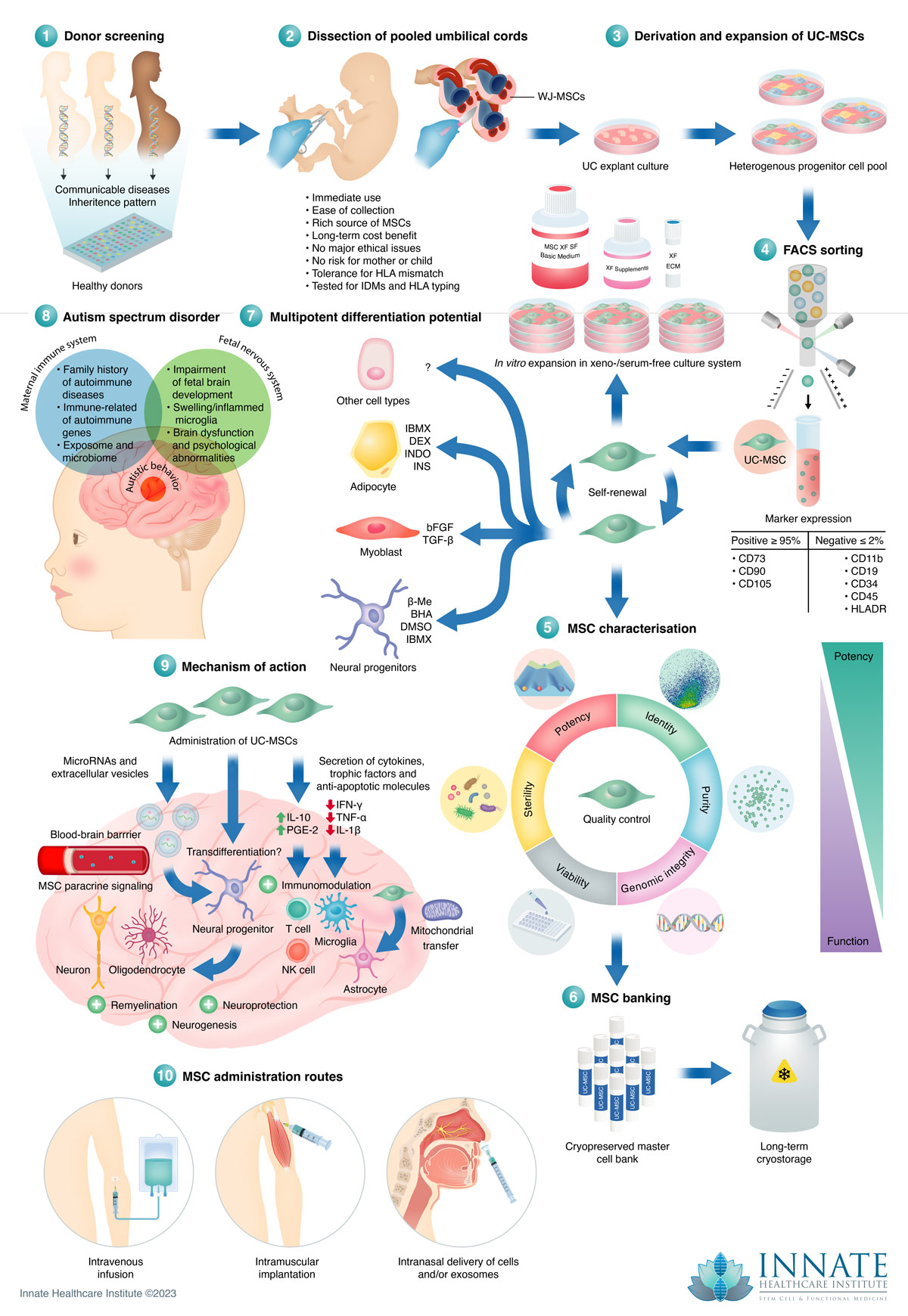
What side effects are there with stem cell therapy for autism?
We are very fortunate to have little to no side effects when it comes to treating autism in children with stem cell therapy. We have NEVER had any moderate or severe side effects, including hospitalizations, immune reactions, graft versus host disease, or major illnesses after treatment. In our experience around 10% of children will either get a little more hyper or a little more tired for the rest of the day after treatment. That is the extent to any side effects that we see. Obvious side effects include bruising or bleeding at the sight of injections. We can’t answer if there will be any “unknown long term” events, for obvious reasons. In our research and experience there’s been no recorded adverse events from UC-MSC treatments that has occurred years after a treatment that is solely due to the UC-MSCs. After decades of use UC-MSC treatments have proven to be incredibly safe when performed properly. And yes, we perform the treatments properly.
What kind of stem cells do you use?
We use umbilical cord tissue derived mesenchymal stem cells (UC-MSC). We believe these cells are superior to other sources of cells, such as bone marrow, adipose, or umbilical cord blood. These cells have several benefits to them, namely that they are donated so there is no need for a surgical procedure to extract them from the child’s body as you would with bone marrow or adipose. Umbilical cord tissue is unique in that there is no severe rejection from the host’s immune system. Making them a very safe and viable option for stem cell treatments.
How does the treatment process work?
The majority of our stem cell treatments for autism are done in the same manner. We do a combination of intramuscular injections (IM), which is just the same as a shot in the shoulder, or like getting a vaccine shot, an intravenous injection (IV), which is usually just on one day, and an intranasal administration. The intranasal (IN) administration is where we take a small device, called a Mucosal Atomization Device, and apply it to the end of the syringe. This is a small, bell-shaped, soft device that gets inserted into the child’s nose and turns the liquid into a spray. The cells are then administered into a specific part of the nose (the sphenopalatine ganglion) where there are an abundance of blood vessels and nerves that can deliver the cells to the brain.
How many days does the treatment take?
The treatments usually take three days for most children. We have found more success in administering the doses multiple times instead of one large dose. We take the total amount of cells that we are going to use and divide it up to be used over three days. For instance, if we’re treating with a total of 300 million cells for the child, then each day 100 million cells will be used over a 3 day period. Each day the treatment is similar. Bigger children can have larger doses so they may require less days of treatment. For example a total treatment dose of 300 million UC-MSCs can be divided into two treatments of 150 million UC-MSCs over two days instead of 100 million UC-MSCs over 3 days. The treatment on the first day lasts a little longer since there is usually more talking on the first day. We like to let the child get accustomed to the clinic and as comfortable as possible. There are toys and a TV for them to watch their favorite shows. This helps with future treatments, so the child isn’t as anxious or fearful of the clinic.
How is your clinic different from other clinics?
Our clinic focuses on a wide range of comprehensive treatments and testing to solve the complex issues of Autism. We’ve noticed many clinics overseas only focus on stem cell therapy, so there’s no follow-ups or other forms of care for the child. Other clinics will only administer the cells IV (see above). Since we don’t focus on what other clinics are doing we can’t answer 100% on how we compare to clinic “X”. We administer higher doses of UC-MSCs on average and focus on other modalities such as GI health, detoxification pathways, heavy metal toxicity, and infections to give a comprehensive approach to healing. Also, we use real UC-MSCs whereas many clinics in the U.S. use stem cell products. These “watered down” versions of stem cells contain healing properties but are not true stem cell treatments like those seen in the research on stem cells and Autism.
How does payment work?
As of October 5th, 2023, our payment protocols have been updated. We require a 50% deposit to schedule the appointment and start preparing for the first treatment. The remaining 50% will be due prior to receiving treatment when you arrive at the clinic on the first day of the treatment series. For patients who would like to explore financing options, we recommend using Advanced Care Card services at www.advancedcarecard.com
The website will prompt you to “Apply Now” and redirect you to their processing website. There, you will be asked a series of questions regarding current financial status and offered a loan agreement with the dollar amount approved and the interest (if any) applied to the loan. If the patient is satisfied with the terms, they will receive a credit card in the mail 7-14 business days after the loan was accepted that can be used towards medical expenses.
Is there anything we need to do to prepare for the treatment?
Yes. We like to make sure every child is as healthy as possible. This can be difficult for us since most families are receiving some sort of additional care from other providers. Some children are on special diets and/or supplements, and we encourage you to continue these. If there is an appropriate need for us to mail any labs to be done at home we can arrange that after the down deposit is collected. Since it can typically take four to six weeks before the first appointment, we will typically send out labs that can be done at home and mailed off before the first treatment is done. These can include labs such as a stool sample, urine sample, or blood spot test. Otherwise, continue any therapies given by other providers.
What medications or supplements do we need to stop taking before SCT?
Currently there are no supplements that are needed to stop being taken before stem cell therapy. Please consult our clinic if you have high dose NSAID or anti inflammatory dependence (examples: Ibuprofen, aspirin, plavix, prednisone, prednisolone) or are using immunosuppressant medications (General: Steroids (Prednisone), Methotrexate, etc.) or Anti-adaptive and anti-innate immunity drugs. Generally, most natural medicines are good for the stem cell therapy.
Do you provide supplements or pharmaceuticals?
We do provide some general supplementation that we see benefit children with autism. In addition, we provide targeted medicines and supplementation for each child’s unique situation. For instance, some children are more hyperactive than others, or may have more GI complications, so we use specific medicine for these children that other children wouldn’t get. We also provide links to continue ordering these medicines at a wholesale cost should they prove to be beneficial. Anyone with children knows that they can be picky when it comes to diet and supplements, and this can be especially true in Autistic children. We have a wide range of medicines, prescription and natural, on hand since you never know what a child will take. We lean towards natural treatment options and keep stock of medicines in liquid and powder forms to accommodate the children better.
Can we talk to other patients you’ve treated?
We truly understand the need to talk to other patients or families treated here, however we do not give out our patients’ or families’ contact information. To better assist, we’ve created a private Facebook Group for familes treated here where we do allow potential parents into the group to ask questions. Please understand that most families are very busy, but they do go this extra step to make you feel as comfortable as possible in sharing their own experience. We do not incentivize patients to tell other people to come to our clinic, there the parents are free to discuss anything good or bad with others regarding our clinic.
What is your success rate for stem cell therapy treatments with autism?
We estimate to have around a 90% success rate with seen improvements in children after stem cell treatments. These successes can vary depending on a variety of factors. But the vast majority of the children we treat see a wide range of changes within the first few weeks to months. Some symptoms we’ve noticed improve earlier than others. Improvements in sleep, behavior, attention, listening, tantrums, and socializing are some of the symptoms to change first. Verbal skills and improvements in speech take a little longer.
How long until we can start treatments?
Most of our first appointments take somewhere around four to six weeks to be scheduled. We’re working very hard to get as many patients treated as possible. If we can get you in for an earlier appointment, we will certainly do so.
How are you sure this will help our child?
We are very confident in our medicine and our experience. We are never 100% sure that this will help a child and we never tell parents that this will be a miracle cure. However, based on previous research and our clinical experience we are very confident it will help most children with Autism. Our testimonials and results speak for themselves.
How many treatments are needed?
In our experience 3-4 treatments are typically needed over the course of a year. 99% of children will start showing improvements after the first and second treatments. Symptoms such as non-verbal will take time to start developing the capabilities to talk and communicate appropriately. Since every child is unique, it is hard to determine exactly how many treatments are needed. However, we have heard parents report seeing improvements over the first 3 months but sometimes these improvements slow down around 6 months. In these instances, the children are typically only treated with 1 low-dose treatment of stem cells and do not follow our diet and supplement guidelines. It is important to continue the therapies we recommend. While we have seen incredible improvements it is important to remember this is not a ‘miracle cure’ that will ‘cure’ Autism after 1 treatment. Most children that show the biggest improvements do so after multiple treatments in addition to undergoing diet and supplement regimen.
Where do you get your stem cells from?
We are unique in that we do not order stem cells from a commercial, third party lab. We harvest umbilical cord tissue derived mesenchymal stem cells from umbilical cords that we buy ourselves. These umbilical cords come from cord banks and or hospitals. They are screened for safety before they are donated to the cord bank or hospital, and they are screened for safety before we process them for the mesenchymal stem cells. The cells are prepared on-site at the clinic before they are administered.
Do we need our own umbilical cord for treatment?
No, you do not need to have your own umbilical cord saved in order to get umbilical cord derived mesenchymal stem cell therapy. At the time of writing this we have only used stem cells from donated umbilical cords and have seen significant improvements.
Does there need to be a compatibility test done with my child and the cells being used?
No. One of the beautiful benefits of using umbilical cord tissue derived mesenchymal stem cells is that they are very ‘universal’. We use these cells for a variety of conditions with no signs of rejection or illness.
How do you know the stem cells are safe to administer?
These cells are karyotyped (chromosomes are counted in these cells). Each cell must have 46 chromosomes. All cells are discarded with any deviations from this number.
Genome stability is very important for cells to proliferate/grow in a steady state. The food/media used to grow these cells are high quality cGMP growth factors that are like ones found within the umbilical cord naturally.
We never stress our cells to grow more than 1-2 passages. So these cells have all the original potency to repair, regenerate, respond to cytokines and secrete paracrine factors and exosomes with the necessary dosage once inside the body.
The morphology of these cells are elongated and fibroblastic. We make sure all cells in the culture have this typical morphology. All cells are discarded with any changes in cell morphology.
We’re well trained and experienced in what we do. As mentioned earlier, we’ve never had any major incidents. As a parent we realize it’s difficult to not be anxious about safety, but we wouldn’t be doing what we do if it wasn’t safe for children. Our track record for safety is perfect in regard to complications from UC-MSCs. The cords are screened for communicable diseases, such as HIV, Hepatitis, Syphilis, and many more. It’s a very stringent process to make sure the biological material is stored, prepared, transported, and processed safely.
Why don't you use bone marrow, adipose, amniotic stem cells or spinal injections?
In our opinion, umbilical cord tissue derived mesenchymal stem cells or a superior source of stem cell therapy. Using bone marrow or adipose stem cells would require a surgical procedure on the child to extract the cells. There’s also some evidence that cells coming from individuals with an autoimmune disease are not as efficient. Many consider autism to be a form of an autoimmune disease. So, there’s a concern that using cells from the child’s body may not work as well as cells from a donated umbilical cord. We don’t perform intrathecal (injecting into the spinal cord) injections. We feel this is a bit too invasive and we’re not operationally setup to perform that kind of procedure here. We feel multiple intranasal deliveries are very efficient in targeting cognitive and brain development without the need for injecting into the spinal cord. However intrathecal procedures can be very effective when performed properly. We just don’t offer them currently.
How do you know the umbilical cords don’t have any genetic material that would lead to cancer, tumors, or future complications?
There is no evidence showing UC-MSC will cause cancer, tumors or lead to diseases in the future. You should consult your doctor if your child currently has or has had a history of cancer. We do not treat children with a current diagnoses of cancer or have not been cancer free for 1 year.
All UC-MSC we grow are monitored for cell growth every day and they divide every 16-22 hours. If they divide too fast or too slow, the entire cell batch is discarded. Also, the surface they grow is on the best quality biomaterial which will not induce any mutations that could lead to cancer. By default, stem cells curb cancer cells by eliciting an immune response to combat cancer (by activating natural killer populations).
A unique characteristic of UC-MSCs is if they are even slightly prone to any mutations, the cells will repair it immediately, but if there is a lot of mutation in stem cells, they activate programmed cell death called apoptosis (and kill themselves).
As of now, it’s extremely rare for UC-MSC or multi-potent stem cells to become cancerous.
How old/young do they have to be? Is there an optimal time to get treated?
The optimal age to start treatments is suggested to be from 2 to 6 years old. However, we have treated kids older and have still seen very positive gains. Certainly, it could be argued that the sooner the better is more ideal to start addressing any concerns.
Is stem cell therapy for autism FDA approved?
No, stem cell therapy for autism is still considered experimental by the FDA. Most FDA approved treatments are pharmaceutical or surgery based. The FDA is primarily concerned with safety of Foods and Drugs (hence their name) and since stem cell therapy is neither a food nor drug it’s our opinion that the FDA will (or should) have little oversight with stem cell therapies being used as a treatment for autism. The treatments are very safe and it is our understanding that the FDA can not govern how a doctor treats their patients. Therefore if a doctor sees it fit to use UC-MSCs as a treatment option it is up to that doctor and their medical board.
How long will the results last?
It’s impossible to know how long the results will last. However, in our experience we don’t hear of any regression in the vast majority of children. The few instances of regression we’ve heard of seem to come in children that are treated only once with a low dose of cells and didn’t follow our prescribed treatment plan. Therefore, we stress to plan on having multiple treatments done. Once a child starts to gain improvements it’s very unlikely those results will just disappear.
How long does it take to see results?
It can take as early as 24 hours to start seeing improvements. The first symptom we see improvements in is typically sleep, with many children sleeping deeper and longer than usual after the first treatment. Most improvements are seen over the first few months. Improvements in behaviors/tantrums, listening, attention span, socializing, sensory sensitivities and other areas typically happen over the first few months. Improvements in verbal communication seem to take the longest, on the average of 6 months. It is important to plan for multiple treatments, especially if a child is non-verbal.
How long have you been performing these treatments?
We’d love to hear from you.
Don’t see your question? Click the button below to contact us. If you prefer immediate assistance, please call us at 602-603-3118.
